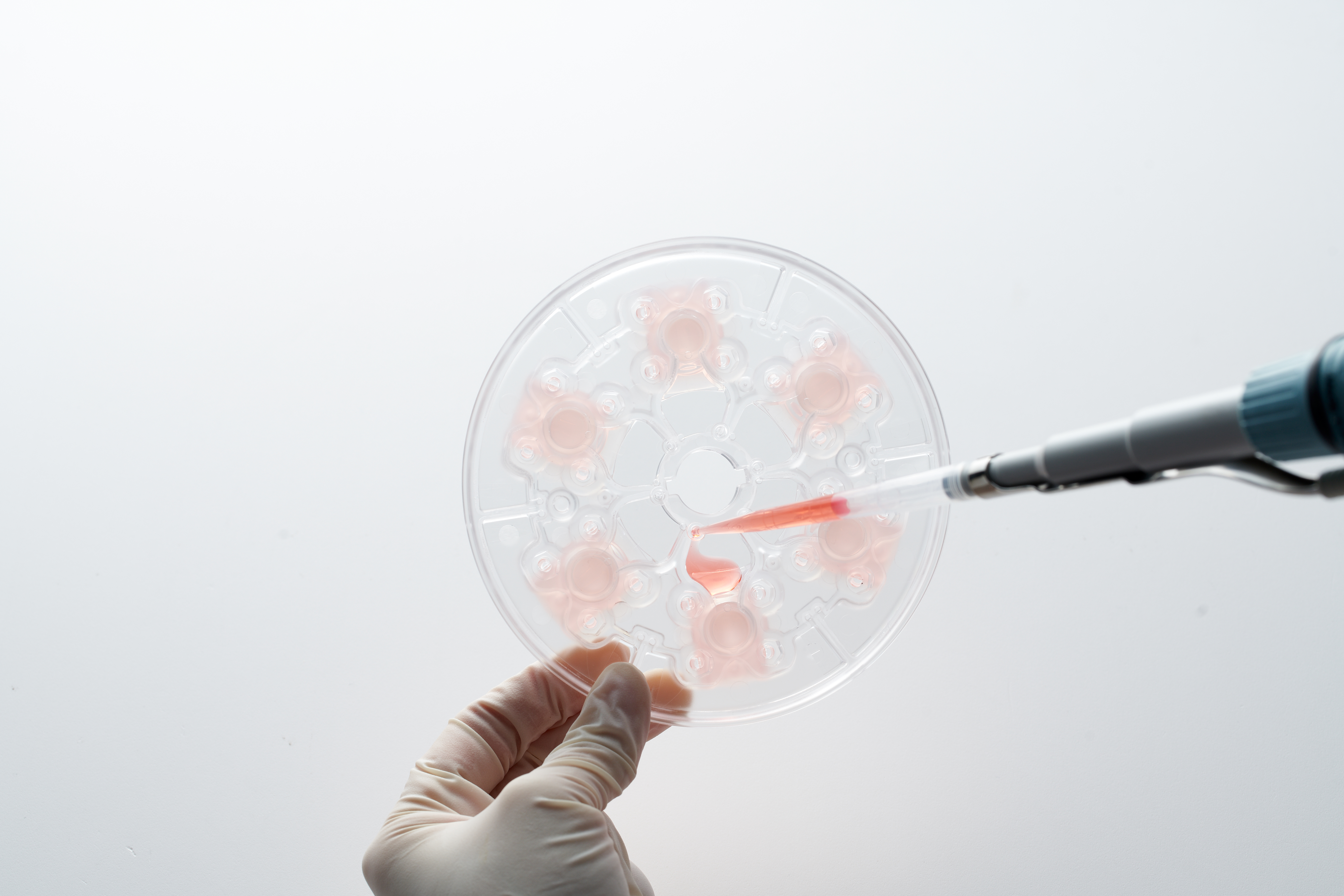
ExoDisc for Rapid, Size-Based Exosome Isolation and Protein Biomarker Analysis
Woo HK, Sunkara V, Park J, et al.
Original ExoDisc paper demonstrating centrifugal microfluidic EV isolation from biosamples with ~95% recovery. Established the technical foundation for the platform.
View Paper (opens in new tab)